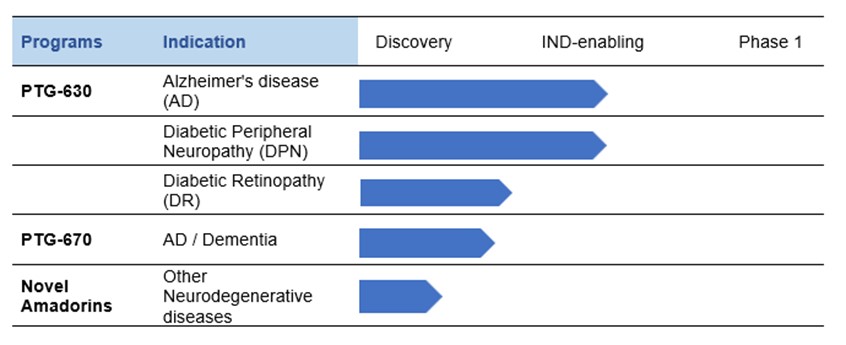
Praetego is currently developing Amadorin candidate PTG-630 as an early intervention to slow or prevent progression of Alzheimer’s disease. This effort is complemented by our program in treating Diabetic Peripheral Neuropathy and Diabetic Retinopathy. A second Amadorin, PTG-670, is under development for AD and Dementia.
Praetego’s proprietary small molecules, the ‘Amadorins’, are are potent inhibitors of the oxidative breakdown of proteins and lipids modified by glucose. This non-enzymatic process forms byproducts called Advanced Glycation End-products, or AGEs. AGE formation and accumulation drive a cascade of damage affecting the vascular system and the central and peripheral nervous systems. Long- term consequences of the AGE pathway are neurodegenerative diseases and serious diabetic complications. AGE pathology is implicated in the chronic diseases of aging.
The Amadorins have been developed and tested over 4 generations. Novel candidates under development offer the greatest potency and margin of safety in nonclinical testing. Praetego has an issued patent for the third generation of Amadorins and patent protection pending for current class of candidates under development (fourth generation).
The original Amadorin demonstrated clinical efficacy against Diabetic Kidney Disease.
Lead Candidate PTG-630
In preclinical safety studies, PTG-630 demonstrated a wide therapeutic window and a minimal risk of drug-drug interactions. It is demonstrating good bioavailability and is well distributed in tissue. In preclinical efficacy studies, PTG-630 has consistently demonstrated protection of both the Central Nervous System (CNS) and the Peripheral Nervous System (PNS) in rodent models of Alzheimer’s Diseases and Diabetic Peripheral Neuropathy. The data demonstrates functional benefits in both models that have been confirmed by direct biochemical measures of neurodegeneration.
Alzheimer’s Disease (AD)
Alzheimer’s disease is a tremendous burden on patients, caregivers, and our healthcare system. A disease limiting agent is critical to ameliorating this devastating disease. In multiple preclinical models of AD, PTG-630 is protecting memory and demonstrating other neuroprotective features. Intended as an oral candidate, it is highly absorbed, crosses the blood-brain barrier and permeates brain tissue.
This work is funded by the National Institute of Aging (NIA) of the NIH.
Diabetic Peripheral Neuropathy (DPN)
Diabetic Peripheral Neuropathy (DPN)—a degenerative process of the nerves, typically starts in the extremities. Affecting eventually approximately 50% of diabetics, DPN can be very painful and debilitating. It can also lead to foot ulcers and amputations.
In proof-of-concept studies using a standard animal model of DPN, several Amadorins tested restored nerve function. They reversed nerve deficits in both large and small nerve fibers. Across the Amadorins evaluated, we confirmed that stronger AGE inhibition led to greater potency in the animals. PTG-630 is demonstrating neuroprotective and neurorestorative effects in the animal model of DPN.
This work is funded by the National Institute of Diabetic and Digestive and Kidney Diseases (NIDDK) of the NIH.
Diabetic Retinopathy (DR)
Diabetic Retinopathy is a leading cause of blindness. Starting as a neurovascular degenerative process, DR leads to vascular damage to the retina and loss of vision. The retina serves as a critical connection of visual data and neural processing. Current treatments for DR include intravitreal injections to inhibit further vascular damage. While these eye injections are moderately effective, they are initiated after substantial irreversible damage has occurred. As oral candidates, the Amadorins are designed to be initiated prior to significant retinal compromise, thus preserving visual function. Preliminary data suggest PTG-630 will be a valuable oral agent against DR.